This manual guides students through essential chemistry experiments, mirroring real-world problem-solving, as seen in MIT’s 1986 approach. It complements texts like LLP, fostering practical skills.
Purpose of a Lab Manual
A chemistry lab manual serves as a structured guide for experimental learning, bridging theoretical knowledge with practical application. It details procedures, safety protocols, and data recording methods, crucial for accurate results. As exemplified by East Los Angeles College’s approach, manuals promote inquiry and environmental awareness.
They ensure consistency, enabling reproducible experiments and fostering critical thinking. Furthermore, manuals, like those accompanying “Principles of General Chemistry,” aid in understanding complex concepts through hands-on experience, preparing students for advanced studies and research.
Importance of Safety in the Chemistry Lab
Safety is paramount in any chemistry laboratory, and a lab manual explicitly outlines crucial protocols. Handling chemicals – acids, bases, and flammable materials – demands strict adherence to guidelines, minimizing risks of accidents and exposure. Proper waste disposal procedures, detailed within the manual, protect both individuals and the environment.
East Los Angeles College emphasizes chemical safety, highlighting its foundational role. Understanding and implementing these precautions, as detailed in Safety Data Sheets (SDS), is essential for responsible experimentation and a secure learning environment.

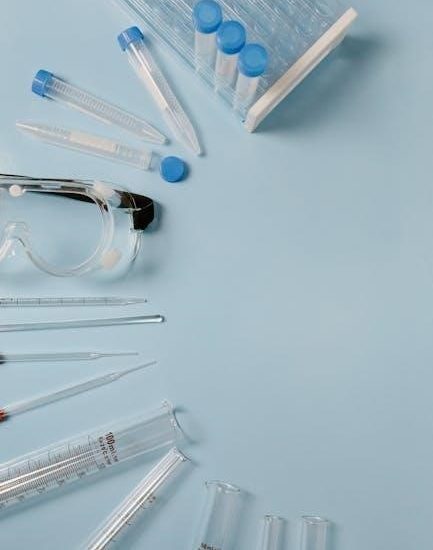
Essential Laboratory Equipment
A well-equipped lab includes glassware (beakers, flasks), precise measuring devices (burettes, pipettes), and tools for controlled heating and stirring, vital for experiments;
Glassware: Beakers, Flasks, and Test Tubes
Beakers are versatile, cylindrical containers for mixing and heating, though not for precise measurements. Flasks, particularly Erlenmeyer flasks, are ideal for titrations and solutions requiring swirling due to their tapered shape, minimizing spillage.
Test tubes, essential for small-scale reactions and qualitative analysis, require careful handling due to fragility. Understanding the specific applications of each glassware type – from general purpose beakers to specialized flasks – is crucial for safe and accurate experimentation. Proper selection ensures optimal results and minimizes risks within the chemistry laboratory.
Measuring Devices: Burettes, Pipettes, and Graduated Cylinders
Burettes deliver precise volumes of liquid, crucial for titrations, utilizing a stopcock for accurate control. Pipettes, available in various sizes, transfer specific volumes with high accuracy, often used for reagent dispensing. Graduated cylinders provide approximate volume measurements, suitable for less demanding applications.
Mastering these devices is fundamental; accurate readings and proper technique are paramount. Each instrument’s precision dictates its use, ensuring reliable data collection and minimizing experimental error in quantitative analysis.
Heating and Stirring Equipment
Hot plates provide controlled and even heating, essential for reactions requiring sustained temperatures. Bunsen burners offer a higher intensity heat source, useful for rapid heating but demanding careful control. Magnetic stirrers utilize a rotating magnetic field to mix solutions, ensuring homogeneity.
Proper equipment selection is vital; consider reaction scale, flammability, and desired temperature. Consistent stirring promotes efficient reactions, while controlled heating prevents decomposition or unwanted side reactions.

Basic Laboratory Techniques
Mastering core techniques – filtration, titration, and heating – is crucial for successful experimentation, building upon foundational skills learned during initial lab sessions.
Filtration Techniques
Filtration is a fundamental separation method utilized to isolate solids from liquids or gases. This process relies on a filter medium, allowing the passage of fluids while retaining particulate matter; Different filtration types exist, including gravity filtration, employing the force of gravity, and vacuum filtration, accelerating the process with reduced pressure.
Proper technique involves selecting appropriate filter paper, ensuring a secure setup to prevent spills, and carefully pouring the mixture to avoid disturbing the collected solid. Understanding filtration is vital for purification and analysis in chemistry;
Titration Procedures
Titration is a quantitative analytical technique used to determine the concentration of an unknown solution, known as the analyte. This involves reacting the analyte with a solution of known concentration, the titrant, until the reaction is complete, indicated by an endpoint.
Accurate titration requires careful standardization of the titrant, precise volume measurements using burettes, and appropriate endpoint detection methods, often employing indicators. Mastering titration is crucial for quantitative analysis in chemistry.
Heating and Evaporation Methods
Heating and evaporation are fundamental techniques for separating mixtures and concentrating solutions in chemistry. Methods include using hot plates, Bunsen burners (with caution!), and steam baths for controlled heating. Evaporation allows solvent removal, leaving behind the solute.
Proper technique involves gentle heating to prevent splattering, utilizing appropriate glassware, and understanding boiling points. These skills are vital for synthesis and purification processes, as highlighted in organic chemistry labs.

Chemical Safety Protocols
Prioritizing safety is crucial; East Los Angeles College’s manual emphasizes it. Proper handling of chemicals, waste disposal, and understanding SDS information are paramount for a safe lab environment.
Handling Acids and Bases
Acids and bases require meticulous handling due to their corrosive nature. Always wear appropriate personal protective equipment (PPE), including gloves, safety goggles, and a lab coat, to prevent skin and eye contact. When diluting acids, always add acid to water, never water to acid, to avoid violent reactions and potential splattering.
Neutralize spills immediately with appropriate neutralizing agents, following established laboratory protocols. Be aware of the pH scale and the relative strengths of different acids and bases. Proper labeling of all acid and base containers is essential, and incompatible chemicals must be stored separately to prevent accidental mixing and hazardous reactions.
Working with Flammable Materials
Flammable solvents and gases pose significant fire hazards; therefore, extreme caution is paramount. Ensure adequate ventilation is present to prevent the build-up of flammable vapors. Keep these materials away from open flames, sparks, and heat sources. Never work with flammable substances near ignition sources.
Proper grounding and bonding procedures are crucial when transferring flammable liquids to prevent static electricity build-up. Know the location of fire extinguishers and fire blankets, and be trained in their proper use. Store flammable materials in designated, fire-resistant cabinets.
Waste Disposal Procedures
Proper waste disposal is critical for environmental protection and lab safety. Never pour chemicals down the drain unless specifically authorized. Separate waste into designated containers labeled for acids, bases, solvents, and solid waste. Consult Safety Data Sheets (SDS) for specific disposal instructions for each chemical.
Halogenated and non-halogenated solvents must be segregated. Broken glass should be disposed of in designated sharps containers. Always follow your institution’s specific waste disposal guidelines and regulations to ensure responsible chemical handling.

Experimental Design and Data Analysis
Effective experiments begin with a clear hypothesis, followed by meticulous data collection and recording. Analyzing results allows for informed conclusions and validation.
Formulating a Hypothesis
A well-defined hypothesis is crucial for a successful experiment, acting as a testable prediction based on initial observations or existing knowledge. As advisors at MIT would pose problems, students must then formulate a potential answer. This involves identifying variables – independent, dependent, and controlled – and predicting the relationship between them;
The hypothesis should be clear, concise, and falsifiable, meaning it can be proven wrong through experimentation. It guides the experimental design and provides a framework for interpreting the collected data. A strong hypothesis isn’t a guess; it’s an educated prediction based on research and understanding.
Collecting and Recording Data
Accurate data collection is paramount in any chemistry experiment. Observations, both quantitative and qualitative, must be meticulously recorded in a lab notebook or designated data sheet. This includes measurements, color changes, temperature readings, and any unexpected occurrences.
Data should be organized, clearly labeled with units, and recorded immediately to minimize errors. Employ appropriate measuring devices (burettes, pipettes) and techniques. Detailed records allow for thorough analysis and reproducibility, essential for validating experimental results and drawing meaningful conclusions.
Analyzing Experimental Results
Post-experiment, rigorous analysis of collected data is crucial. This involves organizing information, performing calculations, and identifying trends or patterns. Compare experimental results to theoretical predictions, evaluating potential sources of error and their impact on accuracy.
Graphical representations, like charts and graphs, can aid in visualizing data and drawing conclusions. A well-documented analysis, supported by evidence, allows for informed interpretations and a comprehensive understanding of the chemical principles demonstrated.

Common Chemical Reactions
This section details fundamental reaction types – acid-base, redox, and precipitation – essential for understanding chemical transformations explored within laboratory experiments.
Acid-Base Reactions
Acid-base reactions, a cornerstone of chemistry, involve the transfer of protons. Lab experiments will focus on neutralization reactions, titrations utilizing burettes and pH indicators to determine equivalence points. Students will explore strong and weak acids/bases, observing their differing behaviors and calculating reaction stoichiometry.
Understanding these reactions is crucial for quantitative analysis and predicting product formation. Practical applications include determining unknown concentrations and preparing buffer solutions, skills vital for further study.
Redox Reactions
Redox reactions, involving electron transfer, are fundamental to many chemical processes. Laboratory investigations will center on oxidation-reduction titrations, utilizing potentiometry and standardized solutions to quantify redox potentials. Students will identify oxidizing and reducing agents, balancing complex redox equations, and observing color changes indicative of electron flow.
These experiments build a foundation for understanding corrosion, batteries, and metabolic pathways, emphasizing practical applications of redox principles in diverse fields.
Precipitation Reactions
Precipitation reactions, forming insoluble products, are crucial for qualitative and quantitative analysis. Lab work will involve mixing solutions to induce precipitate formation, followed by filtration, washing, and drying to determine the precipitate’s mass. Solubility rules will be applied to predict product formation.
Students will master techniques for identifying ions through selective precipitation, understanding stoichiometry, and minimizing errors in gravimetric analysis, linking theory to practical laboratory skills.

Inorganic Chemistry Experiments
This section details qualitative ion analysis and synthesis of inorganic compounds, building foundational skills in identifying and creating these essential chemical substances.
Qualitative Analysis of Ions
Qualitative analysis focuses on identifying ions present in a sample, rather than determining their quantities. This involves a series of selective precipitation and separation techniques, utilizing differences in solubility. Students will learn to systematically confirm the presence of common cations and anions through characteristic reactions, observing color changes and precipitate formation.
Careful observation and proper reagent addition are crucial for accurate identification. This process builds a strong understanding of ionic behavior and lays the groundwork for more complex analytical methods.
Synthesis of Inorganic Compounds
This section details procedures for creating new inorganic compounds from simpler reactants. Experiments may involve precipitation reactions, carefully controlled heating, and techniques to ensure product purity. Students will learn to balance chemical equations, calculate theoretical yields, and assess percent recovery.
Understanding stoichiometry and reaction conditions is vital for successful synthesis. Emphasis is placed on safe handling of chemicals and proper waste disposal, aligning with established laboratory protocols.

Organic Chemistry Experiments
This section focuses on techniques like distillation and synthesis, crucial for purifying and creating organic molecules, as highlighted in resources for organic chemistry students.
Distillation and Purification Techniques
Distillation, a cornerstone of organic chemistry, separates liquids based on boiling points, vital for achieving purity. This manual details simple, fractional, and vacuum distillation methods, emphasizing proper apparatus setup and careful temperature control.
Purification extends beyond distillation, encompassing recrystallization and extraction. Recrystallization leverages solubility differences, while extraction utilizes solvent partitioning. Mastering these techniques, as emphasized in lab resources, is fundamental for obtaining pure compounds for subsequent analysis and reactions.
Safety protocols, including proper handling of heating mantles and flammable solvents, are paramount throughout these procedures.
Synthesis of Organic Compounds
Organic synthesis forms the heart of many chemistry labs, demanding precise technique and understanding of reaction mechanisms. This section guides students through multi-step syntheses, starting with fundamental reactions like esterification and saponification, building towards more complex molecules.
Detailed procedures outline reagent preparation, reaction conditions (temperature, pressure), and work-up procedures. Emphasis is placed on monitoring reaction progress and employing appropriate purification techniques, like distillation, to isolate the desired product.
Safety considerations regarding handling reactive chemicals and potential hazards are thoroughly addressed.
Spectroscopic Analysis (brief mention)
Spectroscopic techniques are crucial for confirming the structure of synthesized organic compounds. While a full treatment is beyond this manual’s scope, an introduction to key methods is provided. This includes basic interpretation of 1H NMR spectra, identifying characteristic signals and integration patterns.
Infrared (IR) spectroscopy is also briefly covered, focusing on recognizing functional group absorptions. Students will learn how these techniques complement other analytical data, providing a comprehensive assessment of product identity and purity.

Quantitative Analysis
This section details methods for determining the amounts of substances, utilizing gravimetric and volumetric analysis techniques for precise measurements and calculations.
Gravimetric Analysis
Gravimetric analysis involves determining the amount of a substance by isolating it and measuring its mass. This technique relies on converting the analyte into an insoluble form, often a precipitate, which is then carefully filtered, washed, and dried. Accurate weighing is crucial for precise results.
Common applications include determining the chloride content in a sample or analyzing the sulfate concentration. Careful attention to detail, including complete precipitation and proper drying techniques, is essential to minimize errors and ensure reliable quantitative data.
Volumetric Analysis
Volumetric analysis, also known as titration, determines the amount of a substance by reacting it with a solution of known concentration. This method utilizes precise measurements of volumes, typically employing burettes, pipettes, and graduated cylinders for accurate delivery;
Indicators signal the endpoint of the reaction, allowing for stoichiometric calculations to determine the analyte’s concentration. Acid-base titrations and redox titrations are common examples, requiring careful standardization of solutions for reliable quantitative results.

Appendix
This section provides essential reference materials, including common chemical formulas, molecular weights, and crucial Safety Data Sheets (SDS) information for safe handling.
Common Chemical Formulas and Molecular Weights
This appendix details frequently encountered chemical formulas vital for calculations within experiments. Included are essential compounds like water (H2O, 18.015 g/mol), hydrochloric acid (HCl, 36.46 g/mol), and sodium hydroxide (NaOH, 40.00 g/mol).
Further listings cover acids – nitric acid (HNO3, 63.01 g/mol) – and bases, alongside hydrocarbons such as methane (CH4, 16.04 g/mol) and naphthalene (C10H8, 128.19 g/mol).
These values are crucial for stoichiometric calculations, determining reagent quantities, and analyzing experimental results accurately, supporting the practical application of chemical principles.
Safety Data Sheets (SDS) Information
Safety Data Sheets (SDS) are critical resources providing comprehensive information about chemical hazards. Each SDS details physical and chemical properties, potential health effects, safe handling procedures, and emergency protocols.
Understanding SDS is paramount for responsible lab work, especially when handling acids, bases, or flammable materials.
This section emphasizes accessing and interpreting SDS before using any chemical, ensuring awareness of risks and appropriate protective measures, aligning with stringent laboratory safety protocols and regulatory compliance.